|
1/31/2024 0 Comments Carbon metal ion bonding name 
The name doesn't use the hydro something acid notation, so the phosphoric must be referring to the polyatomic ion phosphate. Thus, the acid nomenclature rules are used. The name has the word acid in it therefore, it is an acid. One proton is needed to cancel the charge of the chloride ion, so hydrochloric acid is HCl.Įxample, phosphoric acid. 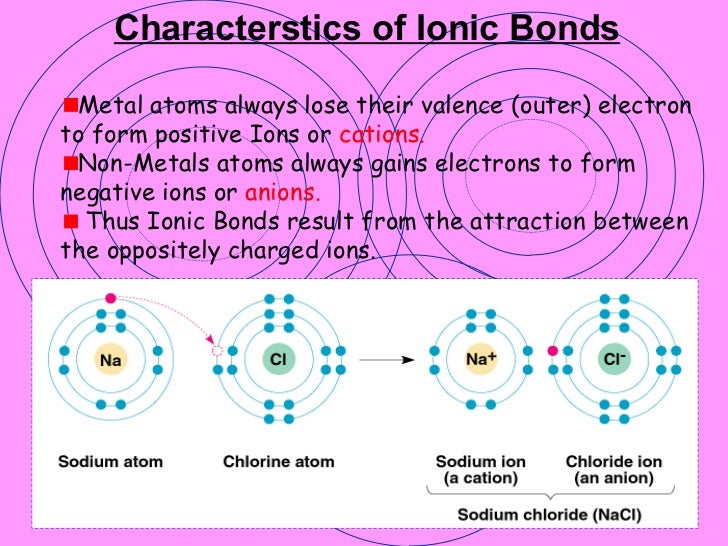
The chloro is referring to the chloride anion, Cl. Only acids based on monoatomic ions use the hydro something acid notation. ( hint: For our purposes, only covalent compounds use prefixes therefore, the presence of a prefix means the compound must be covalent.)Īcids: add as many protons (H +) as needed to cancel the charge of the anion.įor example, hydrochloric acid. Thus, the covalent molecule nomenclature rules are used.Ĭarbon is C, idodide is I, and tetra means 4, so carbon tetraiodide is CI 4. Carbon is a nonmetal, and iodide is a nonmetal therefore, carbon and iodide form a covalent compound. The anion may or may not be polyatomic.Ĭovalent compunds: write the element with a subscript that corresponds to the prefix.įor example, carbon tetraiodide. Covalent compounds are made from two nonmetals. Ionic compounds are made from a metal and a nonmetal. To determine a formula of a compound from its name you have to identify a compound as Ionic Compound
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
